Team Leader

We bring about superproteins.
* Due to the reorganization starting as new centers in April 2018, this laboratory is now belong to the Center for Biosystems Dynamics Research. As for the latest information, please see the following URL below.
> The webpage of Laboratory for Nonnatural Amino Acid Technology, Center for Biosystems Dynamics Research

Team Leader
Kensaku Sakamoto
Room W223, West Building, 1-7-22 Suehiro-cho,Tsurumi-ku, Yokohama 230-0045
Tel: +81-45-503-9459
![]()
What is Synthetic biology?
Proteins useful for life-science studies and medical applications can be synthesized in living cells programmed with the blueprint for the proteins. The cells engineered to utilize novel amino acids in proteins will facilitate the addition of artificial marks onto proteins, and also the synthesis of proteins linked to pharmaceuticals. These cells will help to realize new structures and functions of proteins. We also exploit this platform technology to provide unique, technical supports for drug discovery.
Synthetic Biology should work together with Structural Biology, to develop novel proteins and enzymes based on structural knowledge. Not only for engineering, has such collaboration proved useful for elucidating structural basis for the functions of proteins, receptors, and enzymes.
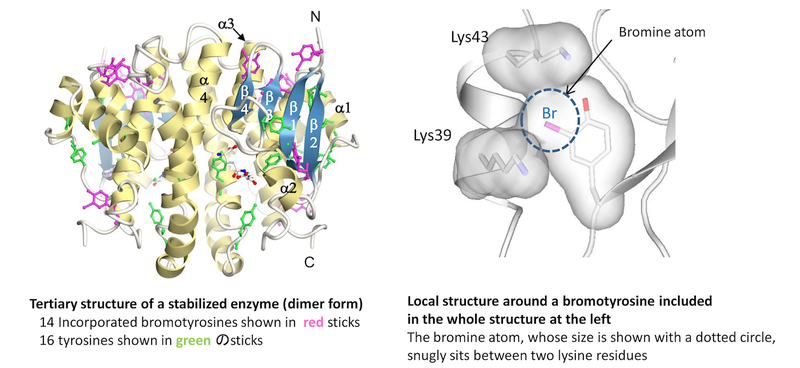




CLST was reorganized into three centers according to the RIKEN 4th Medium-Term Plan from April 1, 2018. For the latest information of Nonnatural Amino Acid Technology Team, please visit the following websites.
> The webpage of Laboratory for Nonnatural Amino Acid Technology, Center for Biosystems Dynamics Research [http://www.bdr.riken.jp/en/research/labs/sakamoto-k/index.html]
> If you continue to browse this site, click here.